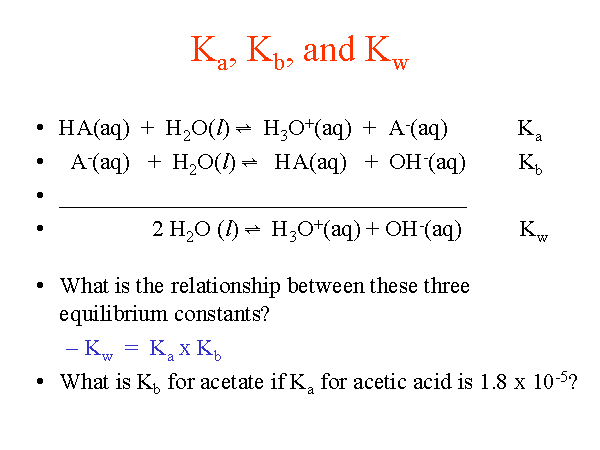Vásárlás: Mitsubishi L 200 2005.12-2015.03 /KA, KB/ Fényszóró bal "dupla fülkés" (H4) sárga indexes (motor nélkül) DEPO 214-1180L-LD-EM Fényszóró árak összehasonlítása, L 200 2005 12 2015 03 KA KB Fényszóró bal dupla

pH, pOH, H3O+, OH-, Kw, Ka, Kb, pKa, and pKb Basic Calculations -Acids and Bases Chemistry Problems - YouTube
What is value of Ka and kb for water (H20) and how numerically prove that ka ×kb=kw in case of water? - Quora

From the dissociation constants Ka and Kb for an acid and its conjugate base, show that Ka· Kb = Kw .
![SOLVED: Helpful Equations pH = -log [HzO+ ] Kw = Ka X Kb [ATJ[Hao+1 [HA] = pOH -log [OH:] 14 = pH + pOH [HA][OH-] [A-] Kb Henderson- Hasselbalch Equation [A-] pH =pKa log - [HA] SOLVED: Helpful Equations pH = -log [HzO+ ] Kw = Ka X Kb [ATJ[Hao+1 [HA] = pOH -log [OH:] 14 = pH + pOH [HA][OH-] [A-] Kb Henderson- Hasselbalch Equation [A-] pH =pKa log - [HA]](https://cdn.numerade.com/ask_images/9f4de4f19628424f8aea53cff3ecadcb.jpg)